Fossil fuels provide a constant and reliable energy supply. The green energy sources need to have the properties. Substitution technologies therefore needs to deliver constant and reliable properties, next its ability to scale to live up to the full energy demand.
What we need: Green Hydrogen and E-Fuels
The world has identified the need to avoid the excavation and substitute the usage of nature destructing fossil fuels. This shall apply to all energy sectors, by clean or preferably green hydrogen or derivate e-fuels.
Hydrogen and two usage routes: Direct use and e-fuels
Usage Route #1 – Hydrogen can either be directly used in industry (e.g. hydrocracking in refineries, production of steel, cement, ammonia) or in new applications in heat and transport (e.g. Fuel Cell Electric Vehicles FCEV). H2 is a highly versatile zero-carbon energy carrier with excellent qualities: it can be burned in heat engines (turbines or combustion engines) and it can be transformed into electricity and heat in fuel cells, zero emission, with only water as exhaust.
Usage Route #2 – H2 can be processed into e-fuels, mostly to be used in existing applications in transport (Figure 2, left bottom). Re-usage of the existing infrastructure for transport & storage is a key argument for these fuels. Their drop-in capability allows for a gradual transition from a fossil into a clean world.
H2-powered fuel cells for applications in transport is about twice as efficient as e-fuels powered combustion engines. Therefore, Route #1 requires only about half of the original green electricity. Methane gas blending in gas networks is a further impactful application of H2
E-Fuels: Power-to-Gas and Power-to-Liquid Technologies
Renewable electricity, unless consumed directly because it concurs with current demand, can be transformed into synthetic fuels for long-term storage and, if needed, for long-range transport.
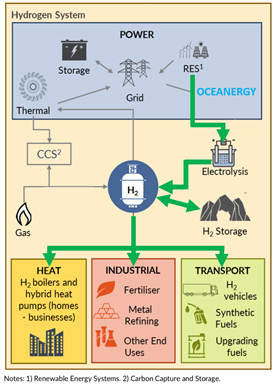
Power-to-gas technologies can transform electricity by electrolysis into hydrogen. As long as we lack a hydrogen infrastructure and because hydrogen is difficult for long-range transport, subsequent methanation or hydrogen-to-liquid technologies may be of advantage. During these processes one adds carbon dioxide (CO2) or nitrogen (N) to hydrogen. The result of methanation is synthetic natural gas – SNG. It is almost pure and clean methane gas. This resembles natural gas, and is even cleaner because SNG does not contain any residual unwanted substances. If one goes the nitrogen route, the result is ammonia (NH3), which is relatively easy to handle, and established technology, also on board of a vessel. It can be liquefied under low pressure or temperature. Ammonia can be used directly in the chemical industry, it can be burned in combustion engines and can be transformed to electricity in special fuel cells. Many think that ammonia will be the main ships fuel of the future.
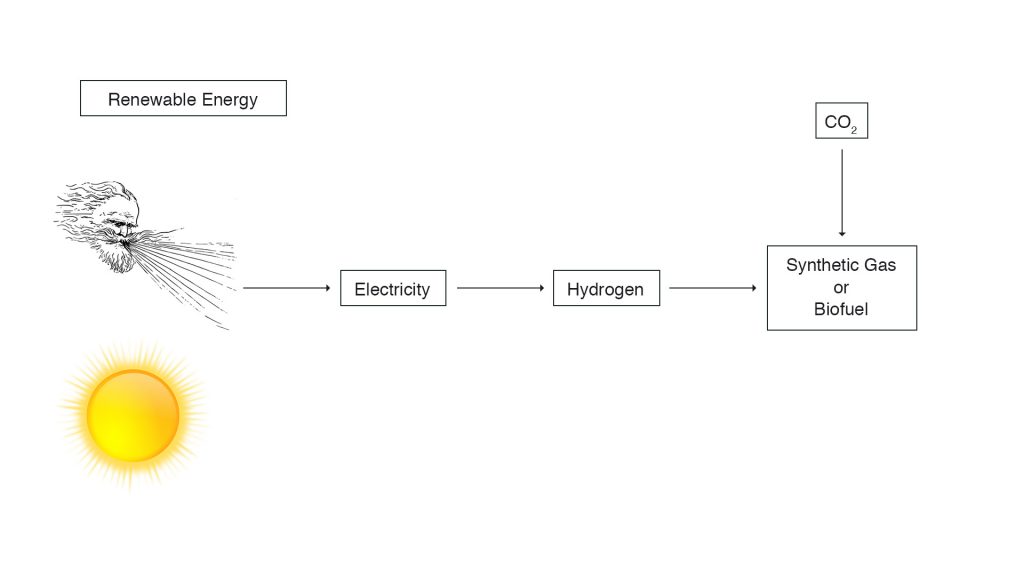
Renewable electricity can also be transformed into synthetic liquid e-fuels. Again, the first step is to produce hydrogen by splitting water H2O into H2 and O2 through electrolysis. Thereafter comes the synthesis of hydrogen via the Fischer-Tropsch process by adding CO and CO2. This process is a collection of chemical reactions that converts a mixture of carbon monoxide, carbon dioxide and hydrogen into liquid hydrocarbons. The result is synCrude, resembling fossil crude oil. From there one can produce a number of hydrocarbon fuels, as diverse as synthetic e-diesel, e-gasoline, e-methanol or e-kerosene.
Substitution technologies in the electricity sector
In the electricity sector there is a substitute, which fulfils these criteria. Nuclear energy can provide constant and reliable electricity. But on the downside it carries the risk of disaster and radioactive pollution of our environment. Further, the safe and long-term disposal of the radioactive waste is not resolved. In any case, nuclear energy can neither cater for cooking and the widespread heating of buildings or water, nor for mobility and transport.
Thermal solar power plants or photovoltaic power plants, combined with short-term storage technologies to get over the night, can deliver constant and reliable electricity in regions with reliable sunshine, such as deserts. So these power plants can bring a local solution for supplying energy within the electricity sector. But then, many of these regions are far away from the hotspots of electricity consumption: The USA, Europe, China and India, and many other places. Therefore electricity production in desert has its limits.
Wind turbines or photovoltaic facilities cannot meet the criteria to deliver constant and reliable energy. Their delivery is intermittent and non-predictable. To be able to replace fossil fuels in the electricity sector one would need to combine their energy production with huge long-term storage capacities. This kind of storage is not available today. Using pump storage power plants would lead to massive nature conservation issues, for instance through building massive dams in mountain regions.
Long-range transportation of electricity from and to areas, where one can suitably produce or store electricity to areas of demand is only a theoretical solution, because of efficiency loss and political dependence, not to speak of the ugly-looking power lines.
Substitution technologies in the heating and mobility sector
Renewable biomass can substitute fossil fuels in the heating and mobility sector to a certain extent. While growing and storing biomass brings a constant and reliable energy supply, it is unfortunately not scalable. Growing biomass for direct burning or for processing into fuels, like for instance ethanol or methane gas, competes directly with the production of food or animal feed. Food prices increase, leading to increased poverty, social unrest and human migration. It is simply unethical to grow fuel on the agricultural fields instead of food, in a world where many people have barely just enough to eat. Biogas from waste is an option but it has only limited capacity.
Substitution technologies for all three energy sectors
Only synthetic fuel gained from renewable energy sources can truly substitute fossil fuels. Fuel contains stored energy, so unlike with electricity from renewable sources there is no storage problem. Transport and distribution does also not pose a problem, it is identical with the transport of fossil fuels today.
Fuels can be in solid, liquid and gaseous form. The latter two are most convenient to handle. Liquid fuels can be transported in tanker ships, tanker trucks or pipelines. Gaseous fuels follow the same principle. Storage is usually only ‘the flip side of the transport coin’ – it happens in tanks and pipe networks. In gas networks, storage can even take place simply by increasing the gas pressure. So often specific gas storage tanks are not even needed.
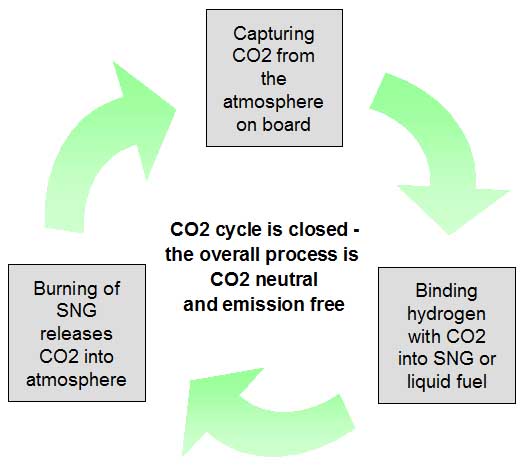
Burning synthetic e-fuels is completely clean, green and CO2 neutral
All synthetic e-fuels have in common that we can burn these in any quantities without remorse and damage to our earth. With synthetic fuels, the CO2 cycle is closed. When burning these fuels, we only release CO2 into the environment, which has been taken out of it before, during the production process. Molecule by molecule. If you want so, the production process resembles the growing process of plants and their photosynthesis, as the theory of man-made greenhouse gases and global warming explains.
Burning hydrogen or e-fuels is clean, it does not smell and it does not harm the climate.
Synthetic e-fuels can be easily stored and transported in existing infrastructures, just like fossil fuels. E-fuels, such as SNG, e-methanol or e-ammonia, are a fully-fledged substitution for fossil fuels. They therefore have the potential to replace fossil fuels, to resolve the problem of increased CO2 concentration in the atmosphere at its root, and therefore to relieve the problem of man-made global warming and unwanted climate change.